Bempedoic Acid Impurities
Bempedoic acid is first-in-class adenosine triphosphate-citrate lyase (ACL) inhibitor used once a day for reducing LDL cholesterol levels in statin-refractory patients. It was developed by Esperion Therapeutics Inc. and approved by the FDA on February 21, 2020. A combination product of bempedoic acid and ezetimibe was approved on February 26, 2020 for increased control of LDL cholesterol levels in patients experiencing refractory elevations despite previous statin treatment. Bempedoic Acid Impurities are primarily used as a reference standard in the pharmaceutical industry for analytical method development and validation to detect and quantify trace levels of this potential contaminant (a nitrosamine impurity) during the manufacturing process of the Bempedoic Acid. It can be used for the analytical method development, method validation (AMV), Quality Controlled (QC) application for Abbreviated New Drug Application (ANDA) or during commercial production of Bempedoic Acid
| Image | Details |
|---|---|
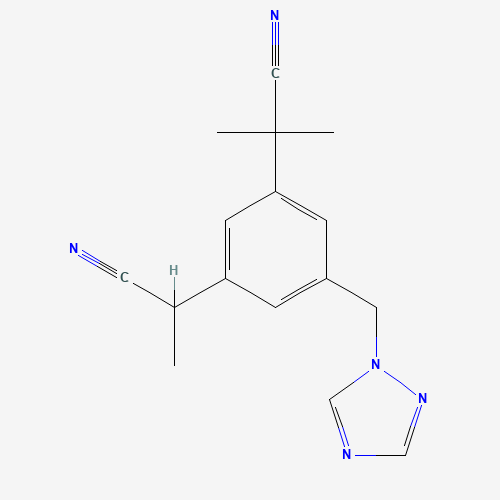 | Impurity Name: Anastrozole Impurity A IUPAC Name: 2,2'-[5-(1H-1,2,4-triazol-1-ylmethyl)-1,3-phenylene]bis(2-methylpropanenitrile) CAS Number: 1215780-15-6 Molecular Formula: C16H17N5 Molecular Weight: 279.34 Status: available |
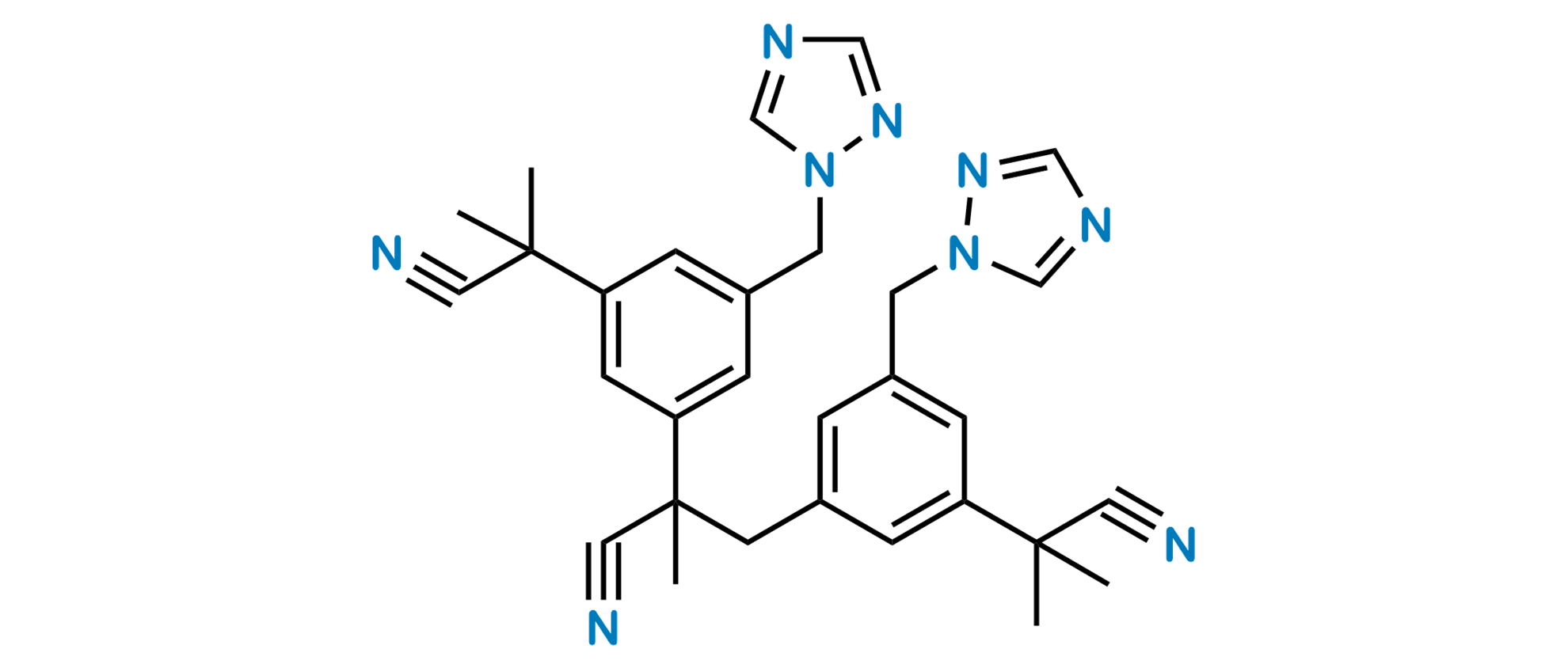 | Impurity Name: Anastrozole Impurity B IUPAC Name: 2,2',2''-[5,5'-(1H-1,2,4-triazol-1-ylmethyl)-1,3-phenylene]tris(2-methylpropanenitrile) CAS Number: 1216898-82-6 Molecular Formula: C30H31N9 Molecular Weight: 517.63 Status: available |
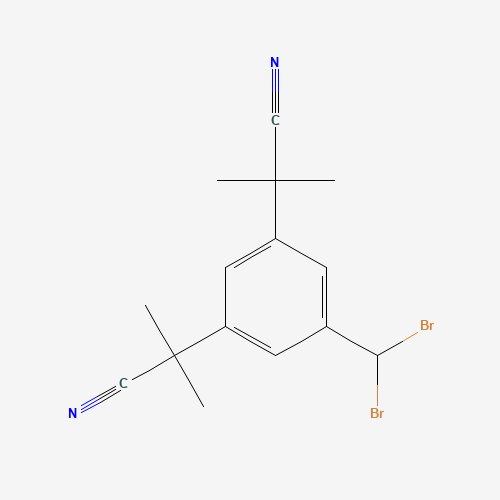 | Impurity Name: Anastrozole Impurity D IUPAC Name: 4,5-dibromo-2-(2-methylpropanenitrile)-1-(2-methylpropanenitrile)benzene CAS Number: 1027160-12-8 Molecular Formula: C15H16Br2N2 Molecular Weight: 384.11 Status: available |
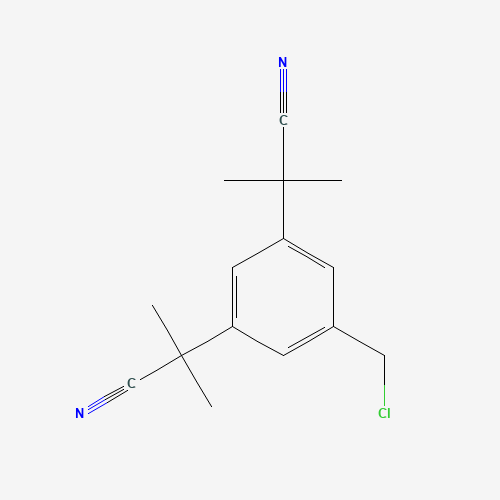 | Impurity Name: Anastrozole Impurity I IUPAC Name: 4-chloro-2,6-bis(2-methylpropanenitrile)benzene CAS Number: 120511-91-3 Molecular Formula: C15H17ClN2 Molecular Weight: 260.76 Status: available |