N-Nitroso Trimethoprim Impurity A
Trimethoprim is an antibiotic. It's used to treat and prevent urinary tract infections (UTIs), such as cystitis. Occasionally, trimethoprim is used to treat other types of infections, such as chest infections and acne. Trimethoprim is an antifolate antibacterial agent that inhibits bacterial dihydrofolate reductase (DHFR), a critical enzyme that catalyzes the formation of tetrahydrofolic acid (THF) - in doing so, it prevents the synthesis of bacterial DNA and ultimately continued bacterial survival. Trimethoprim is often used in combination with sulfamethoxazole due to their complementary and synergistic mechanisms but may be used as a monotherapy in the treatment and/or prophylaxis of urinary tract infections. It is structurally and chemically related to pyrimethamine, another antifolate antimicrobial used in the treatment of plasmodial infections. N-Nitroso Trimethoprim Impurity A is primarily used as a reference standard in the pharmaceutical industry for analytical method development and validation to detect and quantify trace levels of this potential contaminant (a nitrosamine impurity) during the manufacturing process of the drug Trimethoprim. It can be used for the analytical method development, method validation (AMV), Quality Controlled (QC) application for Abbreviated New Drug Application (ANDA) or during commercial production of Trimethoprim.
| Image | Details |
|---|---|
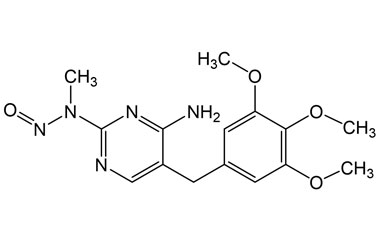 | Impurity Name: Trimethoprim Impurity A IUPAC Name: N-(4-Amino-5-(3,4,5-trimethoxybenzyl)pyrimidin-2-yl)-N-methylnitrous Amide CAS Number: NA Molecular Formula: C15H19N5O4 Molecular Weight: 333.35 Status: available |