Tacrolimus Impurities
Tacrolimus (also FK-506 or Fujimycin) is an immunosuppressive drug whose main use is after organ transplant to reduce the activity of the patient's immune system and so the risk of organ rejection. It is also used in a topical preparation in the treatment of severe atopic dermatitis, severe refractory uveitis after bone marrow transplants, and the skin condition vitiligo. It was discovered in 1984 from the fermentation broth of a Japanese soil sample that contained the bacteria Streptomyces tsukubaensis. Tacrolimus is chemically known as a macrolide. It reduces peptidyl-prolyl isomerase activity by binding to the immunophilin FKBP-12 (FK506 binding protein) creating a new complex. This FKBP12-FK506 complex inhibits calcineurin which inhibits T-lymphocyte signal transduction and IL-2 transcription. Tacrolimus Impurities are primarily used as a reference standard in the pharmaceutical industry for analytical method development and validation to detect and quantify trace levels of this potential contaminant (a nitrosamine impurity) during the manufacturing process of the Tacrolimus. It can be used for the analytical method development, method validation (AMV), Quality Controlled (QC) application for Abbreviated New Drug Application (ANDA) or during commercial production of Tacrolimus.
| Image | Details |
|---|---|
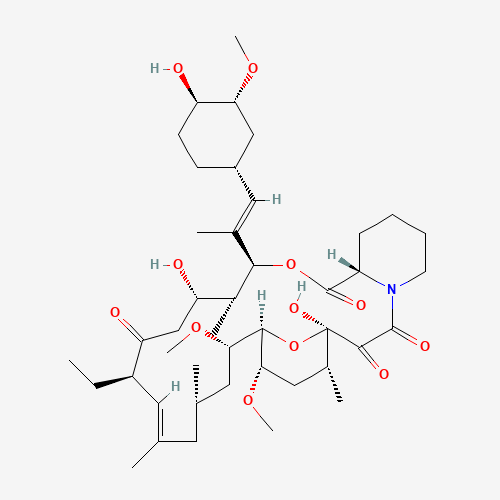 | Impurity Name: Tacrolimus Impurity A IUPAC Name: (1R,3R,4R,6R,7E,9R,10R,12S,13S,14R,15E,17R,18S,19S,21R,23S,24R)-1,3,4,6,9,10,12,13,14,18,19,21,23,24-tetradecahydroxy-7,15-dimethyl-6,10,12,14,18,22-hexaoxo-4,24-epoxytricosa-7,15-diene CAS Number: 104987-12-4 Molecular Formula: C43H69NO12 Molecular Weight: 792.01 Status: available |
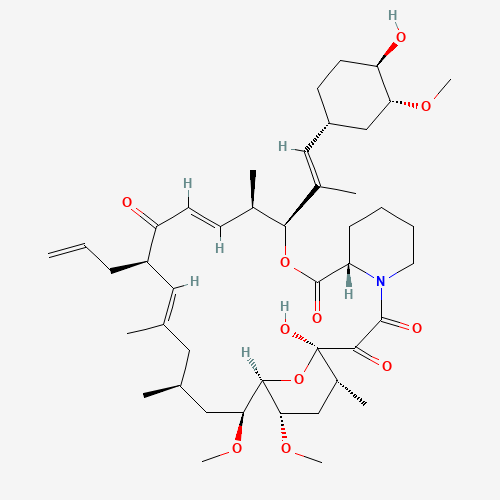 | Impurity Name: Tacrolimus Impurity I / Tacrolimus Diene Impurity IUPAC Name: (1R,3R,4R,6R,7E,9R,10R,12S,13S,14R,15E,17R,18S,19S,21R,23S,24R)-1,3,4,6,9,10,12,13,14,18,19,21,23,24-tetradecahydroxy-7,15-dimethyl-6,10,12,14,18,22-hexaoxo-4,24-epoxytricosa-7,15,17-triene CAS Number: 104987-16-8 Molecular Formula: C44H67NO11 Molecular Weight: 786.00 Status: available |
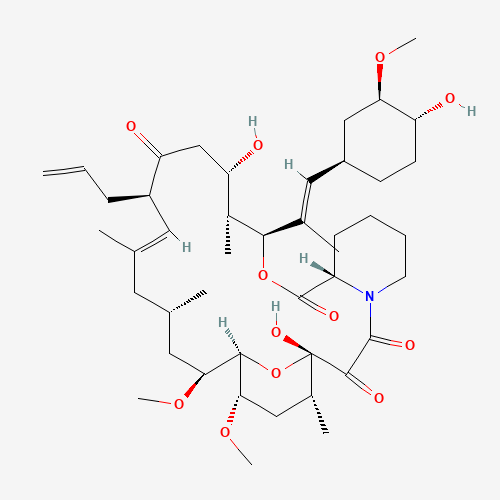 | Impurity Name: Tacrolimus Impurity G / Tacrolimus 19-epimer IUPAC Name: (1R,3R,4R,6R,7E,9R,10R,12S,13S,14R,15E,17R,18S,19R,21R,23S,24R)-1,3,4,6,9,10,12,13,14,18,19,21,23,24-tetradecahydroxy-7,15-dimethyl-6,10,12,14,18,22-hexaoxo-4,24-epoxytricosa-7,15-diene CAS Number: 144490-63-1 Molecular Formula: C44H69NO12 Molecular Weight: 804.02 Status: available |
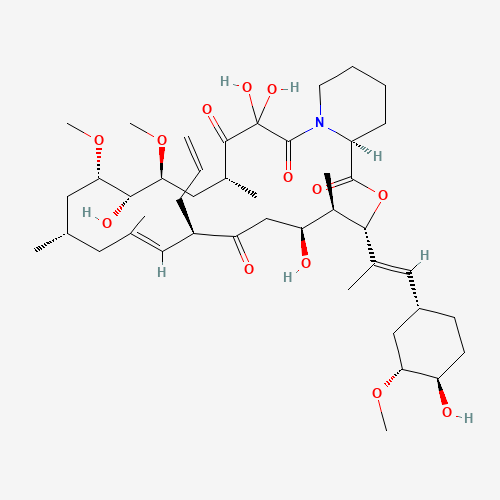 | Impurity Name: Tacrolimus Open Ring Impurity IUPAC Name: (3S,4R,5S,8R,12S,14S,15R,16S,18R,26aS,E)-8-Allyl-5,6,11,12,13,14,15,16,17,18,24,25,26,26a-tetradecahydro-5,15,20,20-tetrahydroxy-3-{(E)-2-[1R,3R,4R)-4-hydroxy-3-methoxycyclohexyl]- 1-methylvinyl}-14,16-dimethoxy-4,10,12,18-tetramethyl-3H-pyrido[2,1-c][1,4]oxaazacyclotricosine-1,7,19,21(4H,8H,20H,23H)-tetrone CAS Number: 144432-23-5 Molecular Formula: C44H71NO13 Molecular Weight: 822.05 Status: available |
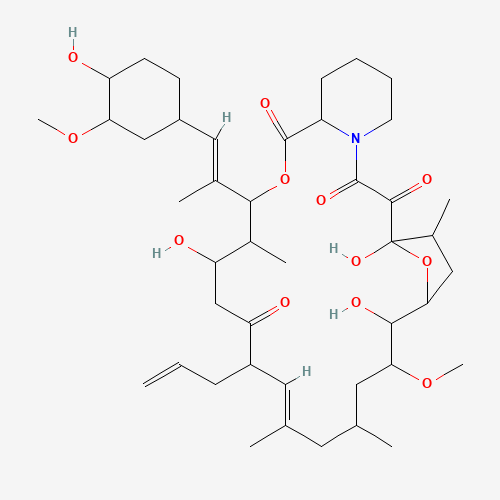 | Impurity Name: Desmethyl Tacrolimus IUPAC Name: (1R,3R,4R,6R,7E,9R,10R,12S,13S,14R,15E,17R,18S,19S,21R,23S,24R)-1,3,4,6,9,10,12,13,14,18,19,21,23,24-tetradecahydroxy-7,15-dimethyl-6,10,12,14,18,22-hexaoxo-4,24-epoxytricosa-7,15-diene CAS Number: 132172-14-6 Molecular Formula: C43H67NO12 Molecular Weight: 789.99 Status: available |